|
Case Report
Neurofibromatosis type 1-associated gastrointestinal stromal tumor: A case report and review of the literature
1 General Surgery Department, São João University Medical Center, Oporto, Portugal
2 Pathology Department, São João University Medical Center, Oporto, Portugal
3 Oncology Department, São João University Medical Center, Oporto, Portugal
Address correspondence to:
Elisabete do Vale Campos
Centro Hospitalar Universitário de São João, Alameda Prof. Hernâni Monteiro, 4200-319 Port,
Portugal
Message to Corresponding Author
Article ID: 100100Z12EC2022
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Campos E, Coelho AR, Freitas MB, Aral M, de Almeida M, Tavares NT, Fonseca E, Melo RB, Barbosa E. Neurofibromatosis type 1-associated gastrointestinal stromal tumor: A case report and review of the literature. J Case Rep Images Surg 2022;8(1):5–10.ABSTRACT
Introduction: Gastrointestinal stromal tumors (GIST), although very rare, are the most common mesenchymal neoplasms of the gastrointestinal tract and develop in approximately 5–25% of patients with neurofibromatosis type 1 (NF1). Neurofibromatosis type 1-associated GIST (NF1-GIST) differ phenotypically and genotypically from sporadic GIST, neither present receptor tyrosine kinase (KIT) or platelet-derived growth factor receptor-alfa (PDGFR-alfa) mutation and have propensity to be multifocal and to occur in the small bowel.
Case Report: We present a case of NF1-GIST, with multiple primary lesions, initially diagnosed as metastatic GIST, with treatment implications.
Conclusion: Different characteristics of NF1-GIST should be empathized so the evidence of multifocal GIST not be confused with advanced/metastatic GIST, influencing treatment options. Identifying this tumor early allows surgical treatment with potential cure, because GIST are mostly treatable tumors with indolent behavior. Currently, no standard drug therapy for unresectable or relapsed NF1-GIST has been established.
Keywords: Case report, Gastrointestinal stromal tumor, Neurofibromatosis type 1
Introduction
Gastrointestinal stromal tumors (GIST) are mesenchymal tumors that arise from the interstitial cells of Cajal of the subepithelial layer of the gastrointestinal tract, which regulate gut motility [1],[2],[3].
Although very rare, with an estimated incidence of 1/100,000/year [4], GIST are the most common mesenchymal neoplasms of the gastrointestinal tract [1],[2].
Some syndromes are linked to GIST, like neurofibromatosis type 1 (NF1) [4]. Patients with NF1 have a higher risk of developing various benign or malignant neoplasms, with GIST developing in approximately 5–25% of patients with NF1 [2],[5].
Active mutations in either the KIT or PDGFR-alfa gene are present in most GIST and they are considered to be the primary drivers of oncogenesis in the disease [2].
Neurofibromatosis type 1 associated GIST (NF1-GIST) cases appear to constitute a subset of wild-type cases, in which neither KIT nor PDGFR-alfa is mutated, implying a different molecular pathogenesis [6]. In NF1-GIST, oncogenesis is attributed to the activation of the RAS/RAF/MAP kinase signaling pathway through loss-of-function NF1 gene mutations [2].
As the oncogenic mechanisms of NF1-GIST are different from those of sporadic GIST, the clinical manifestations also differ. For instance, the median age of onset of NF1-GIST has been reported to be 49 years, compared to 60–65 years for sporadic GIST, and most NF1-GIST develop in the small intestine, especially in the duodenum and jejunum, with multiple lesions [2].
Gastrointestinal stromal tumors association to NF1 is established, but the different aspects of the physiopathological, clinical, and the treatment haven’t been established yet [1], which cause difficulties in the clinical practice.
We present a case of NF1-GIST, with multiple primary lesions, initially diagnosed as metastatic GIST, with treatment implications.
Case Report
We report a case of a 55-year-old female patient, ECOG 0-1, with prior medical history of multiple cutaneous lesions suspected of neurofibromas, pulmonary tuberculosis 10 years ago, cutaneous lymphangioma of thoracic wall and Barlow disease with mitral prolapse and moderate mitral insufficiency medicated with propranolol 40 mg. In a periodic gynecology ultrasound (US), it was detected the pelvic lesions suggestive of neoplasia, the patient had no symptoms.
The pelvic US showed a solid neoformation with 20×16×20 mm superior to the right ovarian and a solid neoformation with 45×31×31 mm in the left adnexal region. Then a pelvic nuclear magnetic resonance (NMR) was made showing multiple solid lobulated lesions in the pelvic region in intime relation with the intestinal wall, independent of ovaries and uterus, probably mesenchymal 3×3 cm in the right and 4×3 cm left, anterior near abdominal wall 2.5×1.9 cm, without adenopathies.
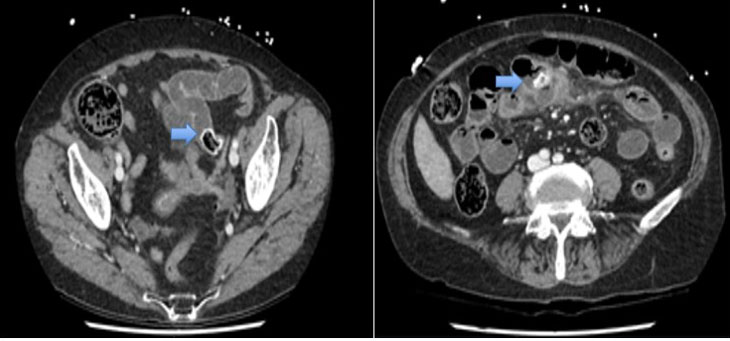
For staging it was ordered a thoraco-abdomin-pelvic (TAP) computed tomography (CT) with evidence of multiple confluent nodular lesions in the pelvic region comprehending an area of 95×75×65 mm, in intime relation with the intestinal wall involving in some locations all the thickness of the wall, three lesions in the left flank in the small intestine wall with 8, 10, and 11 mm, suggestive of neoplastic lesions with intestinal origin, probably neuroendocrine tumor or GIST (Figure 1).
Endoscopy study showed no alterations in the esophagus, stomach, ileum, colon, and lymphangiectasias in the duodenal and proximal jejunum mucosa, without intraluminal lesions.
Positron emission tomography-fluorodeoxyglucose (PET-FDG) confirmed the presence of multiple focus in bowel loops in the pelvic region most evident in the left iliac fossa and hypogastric region (Figure 2).
An ultrasound guided biopsy of one of the pelvic lesions was performed. On histological examination the lesion was composed of a spindle cell proliferation with a syncytial pattern and bland cytology. The mitotic index was low (<1/5 mm2) and there was no necrosis. Immunohistochemical study showed a strong and diffuse staining pattern for DOG1, CD34, and KIT. These findings were compatible with the diagnosis of GIST (Figure 3).
The case was presented in Colo-Rectal Multidisciplinary Tumor Board and it was considered a metastatic GIST and was decided palliative therapy with Imatinib and further molecular study of KIT and PDGFR-alfa.
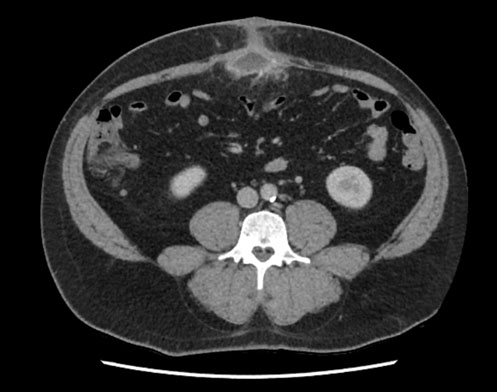
In 3, 6, 9, and 12 months the patient was revaluated with TAP CT with stability of the lesions and no new lesions (Figure 4).
One year after diagnosis and six months after treatment with Imatinib the patient started having symptoms with reference to abdominal pain in the left inferior quadrant but kept regular intestinal transit.
The molecular study was inconclusive for insufficient sample and was repeated showing absence of mutation in genes KIT and PDGFR-alfa, which is associated with resistance to treatment to Imatinib.
The case was presented in Malignant Peritoneal Disease Multidisciplinary Tumor Board and was decided surgical intervention.
During surgical intervention it was detected multiple lesions in the ileum, one lesion in duodenum in D3, one lesion in jejunum 15 cm distal to Treitz angle, and one lesion in the anterior gastric wall, without evidence of carcinomatosis peritoneal or advanced disease. All the lesions were excised and were necessary for an enterectomy of 50 cm long with ileo-ileal anastomosis 40 cm distal to the angle of Treitz (Figure 5). One of the cutaneous lesions was also excised for further study.
Microscopic examination revealed similar morphological and immunohistochemical findings throughout all gastrointestinal lesions (Figure 6). These corresponded to spindle and epithelioid cell proliferations, with a moderate to accentuated cytological atypia, and a low mitotic index (1/5 mm2). The immunohistochemical pattern was compatible with a diagnosis of GIST.
The excised cutaneous lesion corresponded to a neurofibroma (Figure 7). In the post-operation the patient was always hemodynamic stable, with controlled pain but the abdominal pelvis (AP) CT revealed thrombosis of a tributary branch of superior mesenteric vein and it was decided to start hypocoagulation with low molecular weight heparin.
Since there is scarce evidence of survival benefit of Imatinib in patients with NF1-related GIST, it was decided to discontinue treatment with Imatinib and the patient remained in surveillance.
The patient was referred for genetic counseling and the molecular study of NF1 gene confirmed the presence of a pathogenic mutation c.1756_1759delACTA p.(Thr586Valfs*18) in heterozygosity. It was decided to start a high-risk protocol of surveillance regarding the associated risk of central nervous system and breast malignancies and other ocular manifestations of NF1. First-degree relatives were referred for appropriated genetic counseling.
At 12-months post-operative follow-up the patient has no imaging evidence of recurrence.
Discussion
NF1-GIST differ phenotypically and genotypically from sporadic GIST [7]. Studies showed that most NF1-GIST patients had multiple GIST (3 to >100 tumors), ranging from 1 mm to 10 cm in size, mostly small and with a low mitotic rate, predominantly involving the small intestine [7],[8].
The majority of patients were treated surgically and most of them, with long-term follow-up, enjoyed a good prognosis [7],[8].
However, 14% died of metastatic disease, all of these had a tumor >5 cm, mitotic rate >5/50 HPFs, or both. The presence of multiple small tumors was not associated with progressive disease [8].
Although the common indolent behavior of NF1-GIST, there are potentially aggressive forms, more frequent in larger tumors (>10 cm) with high cellularity and mitotic rate >10/50 HPF and coagulative necrosis. In these cases metastasis occurs mostly in the liver and peritoneum, and less frequently to the bone and lung [9].
The standard treatment of localized GIST is complete surgical excision of the lesion, with no dissection of clinically negative lymph nodes (III, A), as lymph node metastasis is extremely rare. If laparoscopic excision is planned, the technique needs to follow the principles of oncological surgery (III, A). A laparoscopic approach is clearly discouraged in patients who have large tumors, because of the risk of tumor rupture, which is associated with a very high risk of relapse. R0 excision is the goal (i.e., an excision whose margins are clear of tumor cells) [4].
Approximately 60% of GIST are cured with surgery alone, especially in cases that involve low-to-moderate risk GIST [10].
Imatinib is the standard treatment for locally advanced inoperable and metastatic disease (I, A), as well as for patients previously treated with adjuvant Imatinib who did not relapse while receiving it. Imatinib is also the standard treatment for patients with metastatic disease who have had all lesions removed surgically, although surgery is not recommended as a primary approach in the metastatic setting [4].
Patients with unresectable or relapsed GIST treated with Imatinib showed an overall response rate exceeding 50% and a median progression-free survival of 1.7 years at a median follow-up time of 10.9 years [11].
In cases of refractory tumors or patients intolerant to Imatinib, Sunitinib, and Regorafenib could be administered as second and third-line treatments, respectively [12].
Mutational analysis is critical to make a clinical decision about adjuvant therapy. The benefit associated of adjuvant Imatinib may vary according to the type of KIT/PDGFR-alfa mutation, being greater in patients with KIT exon 11 deletion mutations. Adjuvant therapy with Imatinib for 3 years is the standard treatment for patients with a significant risk of relapse (I, A) [4].
There is a consensus that PDGFR-alfa D842V-mutated GIST should not be treated with any adjuvant therapy, given the lack of sensitivity of this genotype [4].
With regard to so-called KIT/PDGFR-alfa/BRAF WT GIST, there is a consensus on avoiding adjuvant treatment in NF1-related and SDH expression-negative GIST (IV, D). This reflects their lack of sensitivity to Imatinib and other approved tyrosine kinase inhibitors (TKIs) in the advanced setting, as well as their peculiar natural history, which is often more indolent [4]. Thus, no treatment strategy for unresectable or relapsed NF1-GIST has yet been established [2].
This difference between NF1-GIST and that of the general population has important therapeutic implications. In the presented case the diagnosis of GIST was made prior to the diagnosis of NF1 and so the multifocal presentation was confused with metastatic GIST and treated in conformity. The lack of response to Imatinib, mutational analysis, and NF1 investigation led to another diagnosis which totally change the treatment options and prognosis.
Conclusion
Despite its rarity, providers should consider GIST in patients with a history of NF1 who present with gastrointestinal symptoms or NF1 in patients with GIST with a different phenotype and genotype. Different characteristics of NF1-GIST should be empathized so the evidence of multifocal GIST not to be confused with advanced/metastatic GIST, influencing treatment options. Identifying this tumor early allows surgical treatment with potential cure, because GIST are mostly treatable tumors with indolent behavior. Currently, no standard drug therapy for unresectable or relapsed NF1-GIST has been established. Timely identification of this syndrome enables appropriate genetic counseling of patients and first-degree relatives in addition to the establishment of high-risk surveillance protocols.
REFERENCES
1.
Rahou FZ, Miry A, Bennani A, Bouziane M. Neurofibromatosis type 1 associated multiple and cystic gastrointestinal tumors: 02 case reports. Int J Surg Case Rep 2020;76:210–6. [CrossRef]
[Pubmed]

2.
Fujimi A, Nagamachi Y, Yamauchi N, et al. Gastrointestinal stromal tumor in a patient with neurofibromatosis type 1 that was successfully treated with regorafenib. Intern Med 2019;58(13):1865–70. [CrossRef]
[Pubmed]

3.
Huda T, Singh MP. Gastrointestinal stromal tumors of small intestine. Surg J (N Y) 2019;5(3):e92–5. [CrossRef]
[Pubmed]

4.
Casali PG, Abecassis N, Aro HT, et al. Gastrointestinal stromal tumours: ESMO-EURACAN Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol 2018;29(Suppl 4):iv68–78. [CrossRef]
[Pubmed]

5.
Takazawa Y, Sakurai S, Sakuma Y, et al. Gastrointestinal stromal tumors of neurofibromatosis type I (von Recklinghausen’s disease). Am J Surg Pathol 2005;29(6):755–63. [CrossRef]
[Pubmed]

6.
Valencia E, Saif MW. Neurofibromatosis type 1 and GIST: Is there a correlation? Anticancer Res 2014;34(10):5609–12.
[Pubmed]

7.
Andersson J, Sihto H, Meis-Kindblom JM, Joensuu H, Nupponen N, Kindblom LG. NF1-associated gastrointestinal stromal tumors have unique clinical, phenotypic, and genotypic characteristics. Am J Surg Pathol 2005;29(9):1170–6. [CrossRef]
[Pubmed]

8.
Miettinen M, Fetsch JF, Sobin LH, Lasota J. Gastrointestinal stromal tumors in patients with neurofibromatosis 1: A clinicopathologic and molecular genetic study of 45 cases. Am J Surg Pathol 2006;30(1):90–6. [CrossRef]
[Pubmed]

9.
Omar I, Alsaati H, Waris E. A huge pelvic-abdominal malignant GIST tumour in a patient with neurofibromatosis type 1: Case report and literature review. Case Rep Oncol Med 2020;2020:6590307. [CrossRef]
[Pubmed]

10.
Kalayanamitra R, Shahid Z, Shah N, Patel R, Jain R. The bleeding bowel: A rare case of neurofibromatosis type 1-associated gastrointestinal stromal tumor in a young male. Cureus 2019;11(6):e4868. [CrossRef]
[Pubmed]

11.
Casali PG, Zalcberg J, Le Cesne A, et al. Ten-year progression free and overall survival in patients with unresectable or metastatic GI stromal tumors: Long-term analysis of the European Organisation for Research and Treatment of Cancer, Italian Sarcoma Group, and Australasian Gastrointestinal Trials Group Intergroup Phase III Randomized Trial on Imatinib at Two Dose Levels. J Clin Oncol 2017;35(15):1713–20. [CrossRef]
[Pubmed]

12.
Nishida T, Blay JY, Hirota S, Kitagawa Y, Kang YK. The standard diagnosis, treatment, and follow-up of gastrointestinal stromal tumors based on guidelines. Gastric Cancer 2016;19(1):3–14. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Elisabete do Vale Campos - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ana Rita Coelho - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Marta Baptista Freitas - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Marisa Aral - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Marinho de Almeida - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Nuno Teixeira Tavares - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Elsa Fonseca - Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Renato Bessa Melo - Acquisition of data, Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Elisabete Barbosa - Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2022 Elisabete Campos et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.