|
Case Report
Perianal Paget’s disease regression after treatment for triple negative breast cancer: A case report
1 Resident Medical Officer, Junior Medical Officer Unit, Hunter New England Local Health District, Waratah, NSW, Australia
2 Staff Specialist Pathologist, NSW Health Pathology, John Hunter Hospital, New Lambton Heights, NSW, Australia
3 Consultant General and Breast Surgeon, General Surgery, Calvary Mater Newcastle, Waratah, NSW, Australia
Address correspondence to:
Carissa Vici
Hunter New England JMO Network, Cnr Turton and Tinonee Roads, Waratah, NSW 2298,
Australia
Message to Corresponding Author
Article ID: 100130Z12CV2024
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Vici C, Vilain R, Velaiutham S. Perianal Paget’s disease regression after treatment for triple negative breast cancer: A case report. J Case Rep Images Surg 2024;10(1):10–14.ABSTRACT
Introduction: Extramammary Paget’s disease (EMPD) is a disease affecting apocrine gland-bearing tissue found in the anogenital region, axilla, and other sites. Due to the rarity of this disease, the etiology and pathogenesis are poorly understood. Extramammary Paget’s disease is classified into primary and secondary disease, with secondary EMPD almost always associated with synchronous or metachronous malignancy. The management of secondary perianal EMPD is surgical excision of the area with consideration of radiotherapy and topical or systemic chemotherapy in addition to treating the underlying malignancy.
Case Report: This report describes a patient with partially resected perianal Paget’s disease following an abdominoperineal resection of anal carcinoma. The patient then needed treatment for a metachronous invasive triple-negative breast cancer. Following breast cancer treatment, the perianal Paget’s disease appeared to have regressed.
Conclusion: We report a rare case of perianal extramammary Paget’s disease with metachronous colorectal, then breast cancer responding to treatment with systemic chemotherapy for triple-negative breast cancer.
Keywords: Extramammary Paget’s disease, Perianal Paget’s disease, Spontaneous regression, Triple negative breast cancer
Introduction
Extramammary Paget’s disease (EMPD) is a rare condition with only a few hundred cases reported in the literature worldwide [1]. It affects apocrine gland-bearing tissue, namely the vulva, scrotum, penis, perineum, and anus [1],[2]. Its etiology and pathogenesis are incompletely understood; however, it can be classified into primary and secondary EMPD disease [1],[2],[3]. Primary EMPD develops as an intraepithelial neoplasm within the epidermis, a relatively benign local disease [1]. Secondary EMPD is postulated to arise from either epidermotropic spread or direct extension from an underlying invasive cancer, mainly colorectal and tubo-ovarian cancers [1],[2],[3]. Extramammary Paget’s disease mimics more common skin disorders such as eczema, psoriasis, and fungal skin infections [2]. This often leads to a significant delay in diagnosis which can be confirmed histologically after an excision biopsy [2]. Following the diagnosis of perianal EMPD, investigation for an underlying malignancy is crucial, followed by local treatment of the EMPD [2].
This report details a patient with incompletely resected perianal Paget’s disease, which completely regressed following treatment for her triple-negative breast cancer.
Case Report
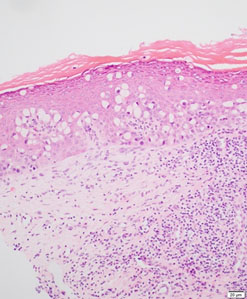
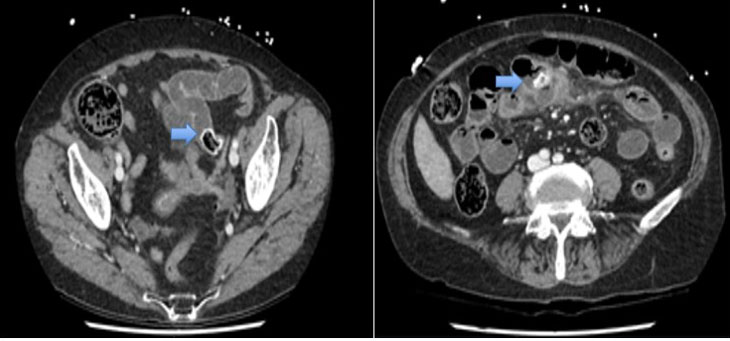
A postmenopausal, asymptomatic Caucasian female in her 50s was diagnosed with anal high-grade mucinous adenocarcinoma following a positive screening fecal occult blood test and colonoscopy. On physical examination at the colonoscopy, a 3 cm non-circumferential anal mass was palpated 0–3 cm from the anal verge located mainly at the posterior bowel wall. The examination was otherwise unremarkable. There is no significant family history of bowel or breast malignancy, and gene testing showed no abnormalities. She then underwent an abdominoperineal resection of the rectum. Histology from this operation revealed a high-grade mucinous adenocarcinoma limited to the submucosa with extensive pagetoid extension along the squamous epithelium to the anal margin (Figure 1). There was no vascular invasion, and the 16 lymph nodes excised were benign. Immunohistochemistry was positive for CDX2, CK20, and CEA and negative for CK7. She had T1N0Mx, Stage 1 rectal cancer and did not require adjuvant therapy.
One year later, at the time of her surveillance colonoscopy, a biopsy of a hyperkeratotic, desquamative skin lesion at the natal cleft on the border of her abdominoperineal resection site was performed. The pathology confirmed residual pagetoid adenocarcinoma in situ showing the same immunohistochemistry profile as the abdominoperineal resection histology (Figure 2, Figure 3, Figure 4). HER2 expression in this specimen was not assessed. A wide local excision of the remaining perianal disease was planned.
While awaiting surgery, the patient was diagnosed with a screen-detected triple-negative invasive high-grade carcinoma of the left breast. She did not have clinical evidence of mammary Paget’s disease. Her case was discussed at the multidisciplinary meeting, and her breast cancer treatment was prioritized over managing the perianal Paget’s disease. The operation for her perianal Paget’s disease was delayed until the completion of her breast cancer treatment. The patient had standard neoadjuvant chemotherapy for triple-negative breast cancer with dose-dense Adriamycin and cyclophosphamide followed by 12 cycles of carboplatin and paclitaxel. Following completion of her neoadjuvant chemotherapy, she had a wide local excision and sentinel node biopsy of the left breast cancer and adjuvant radiotherapy.
After completing her breast cancer treatment, the patient returned to her colorectal surgeon to reschedule the operation to excise the remaining perianal Paget’s disease. However, on examination at this appointment, the perianal lesion was found to have wholly regressed, and surgery was not performed. Two years later, the patient remains well with no recurrence of her perianal Paget’s disease, breast or rectal cancer.
Discussion
Extramammary Paget’s disease is a rare disease that affects areas where apocrine glands are found, such as the vulva, scrotum, penis, and anus [1],[2]. It presents as erythematous lesions that can be painful, pruritic, and oozy and are often misdiagnosed as eczema, fungal infections, and other more common skin conditions [2]. Diagnosis is made by histopathological identification of Paget cells from a skin lesion biopsy [2]. The pathology and staining are similar to that observed with mammary Paget’s disease [4]. While its etiology and pathogenesis are incompletely understood, EMPD can be classified into primary and secondary disease [1],[2],[3]. Primary EMPD is thought to develop from the epidermis as an intraepithelial neoplasm and is associated with a better prognosis [2]. Secondary EMPD is currently understood to develop from either epidermotropic spread or direct invasion from an underlying invasive cancer, mostly colorectal and tubo-ovarian cancers [2].
Given the rarity of EMPD, there are no formal guidelines for its management [1],[2],[5],[6],[7]. However, the first widely accepted step involves detailed investigations to examine for an underlying primary cancer [1]. The current accepted treatment modality is wide local excision of the affected area which is associated with relatively high recurrence rates of 44–60% due to the multicentric nature of EMPD [1]. Moh’s micrographic surgery has been utilized more recently and appears to be associated with lower risk of recurrence [1]. In this case, however, the patient’s perianal EMPD recurrence completely regressed without curative surgery thereby challenging this dogma. Radiotherapy has also been employed for the treatment of EMPD—typically for patients who are unfit for surgery, have experienced recurrence following surgery, for those wishing to avoid a disfiguring surgery and as adjuvant therapy in those with underlying invasive cancer [1]. Chemotherapy has additionally been used in the treatment of EMPD [1]. Topical chemotherapy with 5-fluorouracil, bleomycin, and imiquinoid has been used for relief of symptoms and to reduce disease burden prior to surgery [1]. Systemic chemotherapy with various combinations containing agents such as 5-flurouracil and carboplatin and cisplatin has also been used where both surgery and radiotherapy are contraindicated [1]. Emerging therapies include photodynamic therapy and laser therapy, the latter of which is associated with a very high recurrence rate [1].
Due to the difference in management options and prognosis between primary and secondary EMPD, differentiating between the two is essential and immunohistochemical examination can often assist in confirming the correct diagnosis in most instances. Typical indicators of Paget’s disease concurrent with rectal cancer include a CK20-positive, CDX2-positive, and GCDFP-negative immunophenotype as demonstrated in this case [8]. Conversely, primary anogenital Paget disease often presents a CK7-positive, CK20-negative, and GCDFP-positive immunophenotype [8].
Given her history of rectal cancer, it is more likely that the patient had secondary EMPD. The persistent EMPD on the biopsy from her colonoscopy one-year post-surgery likely represents incomplete resection of the original disease as the pagetoid change seen on the skin biopsy was the same as that seen on the abdominoperineal resection specimen. There have been 2 case reports describing spontaneous regression of EMPD—one following resection of underlying renal cell cancer and one following incomplete resection of primary perianal Paget’s disease; however, this is again a rarely reported phenomenon and is unlikely to account for the regression of perianal Paget’s disease in this case [9],[10].
As spontaneous regression of the patient’s perianal Paget’s disease is unlikely, consideration must be given to the following 2 possibilities—either the perianal Paget’s disease is a paraneoplastic manifestation of the patient’s breast cancer, or the systemic treatment of the breast cancer also treated the concurrent coincidental perianal Paget’s disease.
Firstly, while mammary Paget’s disease is frequently associated with underlying invasive breast cancer, current literature is yet to support a direct link or association between perianal EMPD and underlying breast malignancy [4]. Given the patient’s history of rectal carcinoma and the current understanding of the pathogenesis of secondary EMPD, which involves direct invasion from an underlying invasive cancer, it is unlikely that the patient’s breast cancer is linked to the perianal EMPD. However, a single case has described a patient with synchronous mammary and EMPD of both the breast and vulva [11]. As this is also a rare case and merely coincidental, it could be explained by the formation of Paget’s disease in ectopic mammary tissue along the mammary ridge [11].
It has also been reported that 20–60% of EMPD is associated with the expression of HER2 [2]. Cases identified include perianal, axillary, scrotal, and vulval EMPD, both primary and secondary disease [12]. A high response to systemic chemotherapy with paclitaxel and trastuzumab has been observed in cases of EMPD with HER2 expression which offers the potential for an alternative or additional treatment modality in this subset of patients [2],[12].
Secondly, and more likely, our patient’s systemic treatment for her breast cancer could have concurrently treated her EMPD [1],[2],[4],[5][12]. Systemic agents reported to have been used for the treatment of EMPD with distant metastases include 5-fluorouracil, carboplatin, docetaxel, paclitaxel, and trastuzumab [1],[2],[5],[6],[7]. The patient received both carboplatin and paclitaxel throughout her neoadjuvant course of chemotherapy. Therefore, it is possible that her systemic treatment played a role in the regression of her EMPD.
As this is the first reported case of synchronous perianal EMPD and invasive breast cancer, current literature has not described an association between the two. It remains unclear if the concomitant presence of perianal EMPD has any impact on prognosis. In terms of treatment, while more research is required, the implication of this case could be that for patients with synchronous EMPD and invasive breast cancer, their EMPD can simply be monitored while undergoing standard treatment for their breast cancer with surgery and chemoradiotherapy. After completion of breast cancer treatment, the EMPD could then be reassessed to determine if it has regressed. By adopting this approach, instead of the current accepted first-line curative surgery for EMPD, these patients could potentially avoid a further disfiguring surgery.
Additionally, as discussed above, current options for those who are not fit for, or who do not wish to proceed with, surgery include topical therapies such as imiquimod and 5-fluorouracil or photodynamic therapies; however, these therapies have high incomplete response rates and high recurrence rates [1],[2]. The 5-year survival of EMPD greater than TNM Stage IIIb is only 40%, compared with 80% for those with Stage IIIa or less disease [1]. To date, systemic chemotherapy has only been used in the treatment of EMPD with distant metastases [1]. If systemic chemotherapy is demonstrated to be successful in the treatment of earlier stages of EMPD it could therefore provide an effective cure for those who are not surgical candidates and improve overall survival by treating the disease before it disseminates.
The primary challenge of using systemic chemotherapy for the treatment of EMPD will be determining the subgroup of patients who will receive the most benefit. Systemic chemotherapy is associated with a high side effect profile and those with comorbidities that make them unfit for curative surgery may similarly be unsuited to receiving systemic chemotherapy. Further, in cases of primary EMPD that is localized and relatively indolent, the toxicities of systemic chemotherapy may not outweigh its benefit.
Conclusion
Extramammary Paget’s disease is a rare and incompletely understood disease with only a few hundred cases reported in the literature world-wide. This report details a patient with perianal EMPD that was found to have regressed after the completion of treatment for her invasive triple-negative breast cancer. To our knowledge there have been no documented similar cases. There are three possible suggestions to explain the regression of this patient’s perianal EMPD—spontaneous regression, EMPD as a paraneoplastic manifestation of breast cancer or EMPD responding to systemic breast cancer treatment. While current literature does not support the first two hypotheses, the chemotherapeutic agents administered to this patient for treatment of her breast cancer have been used to treat cases of metastatic EMPD, making it the most likely explanation. Therefore, this case highlights the potential for the use of systemic chemotherapy as an alternative treatment option in patients with EMPD who are not suitable candidates for curative surgery and possibly as a first-line option in those with synchronous EMPD and invasive breast cancer.
REFERENCES
1.
Al Hallak MN, Zouain N. Extramammary perianal Paget’s disease. Case Rep Gastroenterol 2009;3(3):332–7. [CrossRef]
[Pubmed]

2.
Ishizuki S, Nakamura Y. Extramammary Paget’s disease: Diagnosis, pathogenesis, and treatment with focus on recent developments. Curr Oncol 2021;28(4):2969–86. [CrossRef]
[Pubmed]

3.
Noel A, Bhat RM, Rao SV, Fernandes S. An unusual secondary extramammary Paget’s disease of vulva. Indian J Sex Transm Dis AIDS 2020;41(2):210–2. [CrossRef]
[Pubmed]

4.
Lopes Filho LL, Lopes IMRS, Lopes LRS, Enokihara MMSS, Michalany AO, Matsunaga N. Mammary and extramammary Paget’s disease. An Bras Dermatol 2015;90(2):225–31. [CrossRef]
[Pubmed]

5.
Guercio BJ, Iyer G, Kidwai WZ, et al. Treatment of metastatic extramammary Paget disease with combination ipilimumab and nivolumab: A case report. Case Rep Oncol 2021;14(1):430–8. [CrossRef]
[Pubmed]

6.
Fernandes L, Graça J, de Matos LV, et al. Metastatic extramammary Paget disease, a remarkable clinical case and a brief review of a rare disease. Dermatol Reports 2020;12(2):8841. [CrossRef]
[Pubmed]

7.
Stavrou M, Martin L, El-Madani F, Naik V, Papanastasiou S, Gupta S. Perianal Paget’s disease—Report of a rare case. Int J Surg Case Rep 2012;3(10):483–5. [CrossRef]
[Pubmed]

8.
Liao X, Liu X, Fan X, Lai J, Zhang D. Perianal Paget’s disease: A clinicopathological and immunohistochemical study of 13 cases. Diagn Pathol 2020;15(1):29. [CrossRef]
[Pubmed]

9.
Archer CB, Louback JB, MacDonald DM. Spontaneous regression of perianal extramammary Paget’s disease after partial surgical excision. Arch Dermatol 1987;123(3):379–82.
[Pubmed]

10.
Bowling JCR, Powles A, Nasiri N, Searle A, Bunker CB. Spontaneous regression of extramammary Paget’s disease after excision of primary apocrine carcinoma, in an immunosuppressed patient. Br J Dermatol 2005;153(3):676–7. [CrossRef]
[Pubmed]

11.
Popiolek DA, Hajdu SI, Gal D. Synchronous Paget’s disease of the vulva and breast. Gynecol Oncol 1998;71(1):137–40. [CrossRef]
[Pubmed]

12.
Plaza JA, Torres-Cabala C, Ivan D, Prieto VG. HER-2/neu expression in extramammary Paget disease: A clinicopathologic and immunohistochemistry study of 47 cases with and without underlying malignancy. J Cutan Pathol 2009;36(7):729–33. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Carissa Vici - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Ricardo Vilain - Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Shanta Velaiutham - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2024 Carissa Vici et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.