|
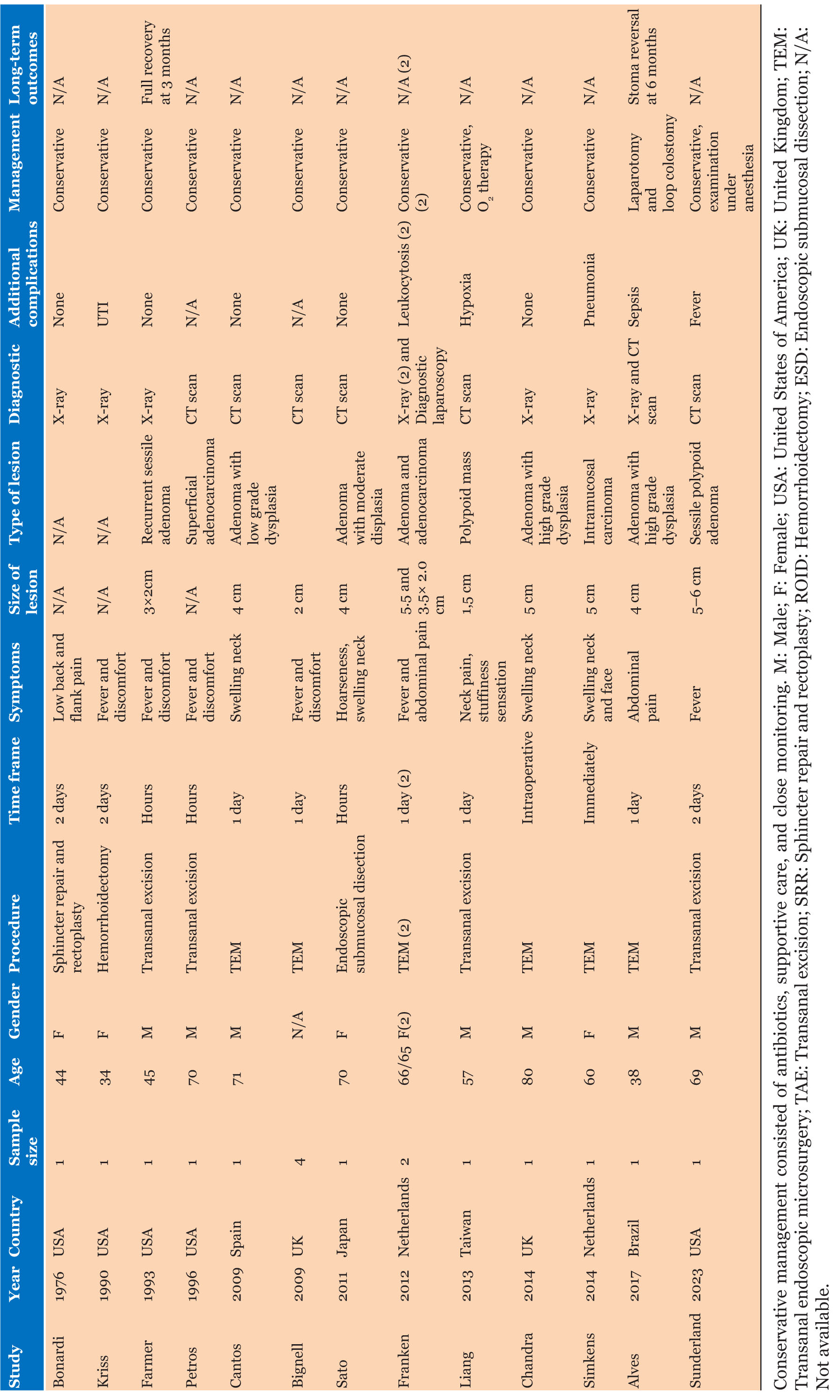
Case Report
Comprehensive treatment and experience of a rare case of gallbladder hepatocellular adenocarcinoma
1 Department of General Surgery, The Second Hospital of Hebei Medical University, Shijiazhuang, Hebei 050000, China
2 Department of General Surgery, Xiongxian Hospital, Baoding, Hebei 071800, China
Address correspondence to:
Miao Tang
Department of General Surgery, The Second Hospital of Hebei Medical University, 215 Hepingxi Road, Shijiazhuang, Hebei Province 050000,
China
Message to Corresponding Author
Article ID: 100155Z12MT2025
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
Tang M, Hu Z, Gao H. Comprehensive treatment and experience of a rare case of gallbladder hepatocellular adenocarcinoma. J Case Rep Images Surg 2025;11(1):37–41.ABSTRACT
Introduction: Hepatoid adenocarcinoma (HAC) is an adenocarcinoma with components similar to those of hepatocellular carcinoma, while HAC of the gallbladder is a rare malignant tumor characterized by diagnostic challenges characterized by diagnostic challenges, high malignancy, and poor prognosis, with only case reports available to date. Surgery is currently the best treatment option. With the advancement of gene sequencing technology, we are gradually able to identify the specific gene mutation types of tumors, providing precise and personalized treatment plans for rare tumors, which is a more rational approach. This case exemplifies the use of gene sequencing to guide the comprehensive treatment of gallbladder HAG, achieving excellent outcomes.
Case Report: The patient is a 58-year-old male who presented with jaundice and abdominal pain as the main symptoms. Laboratory tests indicated a significant increase in alpha-fetoprotein (AFP) and mild anemia. Computed tomography (CT) and magnetic resonance imaging (MRI) revealed a solid tumor in the gallbladder with local tissue infiltration. The patient underwent radical surgery for gallbladder cancer, and the surgically removed tissue was subjected to immunohistochemical staining, which revealed the tumor to be HAC with focal neuroendocrine differentiation. Genetic sequencing showed BRCA2 rearrangement, CDKN2A/B deletion, and TP53 gene mutation in the tumor. After receiving treatment with Tegafur, Gimeracil, and Oteracil Potassium capsules combined with Olaparib, the patient’s AFP progressively decreased, and multiple follow-up examinations showed no new lesions. Thirteen months later, the patient contracted the COVID-19 virus and subsequently died from lung failure.
Conclusion: Surgery combined with gene sequencing technology is feasible for treating primary gallbladder HAC, providing new insights for the comprehensive treatment of rare tumors.
Keywords: BRCA2, Genetic sequence, Hepatoid adenocarcinoma, Olaparib, Rare case
Introduction
Hepatocellular adenocarcinoma (HAC) is the general term for primary extrahepatic tumour of hepatocyte-like differentiation, with or without secretion of alpha-fetoprotein (AFP). Reportedly, 83.9% of HAC occurs in the stomach, while some cases are reported in the lung, kidneys, pancreas, or ovary [1]. However, gallbladder HAC is rare and there are few reports of incidence. It is currently postulated that these organs originate from the anterior intestine during embryonic development, similar to the liver, so some endothelial cells may have the potential for bimodal differentiation. These endothelial cells can differentiate into primitive intestinal epithelial cells but also have a hepatocyte lineage [2]. Alternatively, some studies hypothesize that adenocarcinoma acquires hepatocyte phenotype during tumor development [3],[4].
Since gallbladder HAC has pathological characteristics of both adenocarcinoma and hepatocellular carcinoma, it is often difficult to distinguish clinically from hepatocellular carcinoma, aggressive gallbladder carcinoma, or adenocarcinoma-producing AFP. Concerning histology, HAC tumor cells demonstrate similar morphology to hepatic cells and overlap in immune marker expressions. Hence, gallbladder HAC diagnosis requires a combination of radiology, histology, and immunohistochemistry results.
Gallbladder HAC is highly malignant and invasive. When discovered, it is often accompanied by liver or lymph node metastases, and most patients only survive for a few months (with or without surgery). Surgery is still the best treatment option at present, and no specific drugs have been shown to be effective. Although no authorized guidelines have been provided for HAC, research on the molecular features of HAC has provided direction for the treatment and highlighted new potential targets. Researchers have reported a case of primary gallbladder HAC with MB21D2/GALNT12/ARID2 gene mutations [5], which suggests that the pathogenesis and treatment plan of gallbladder HAC should be special and unique. We treated a patient with gallbladder HAC using surgery and gene sequencing technology for personalized treatment, which effectively extended the patient’s overall survival (OS) and progression-free survival (PFS).
Case Report
The patient, a 58-year-old male, presented with jaundice as the initial symptom. His medical history includes underlying conditions such as hypertension, coronary heart disease, and type 2 diabetes. However, there is no history of abdominal surgery, liver diseases, gallstones, or alcohol consumption. No evident abdominal pain or hepatosplenomegaly was observed during the abdominal examination. The patient denied family inheritance, alcohol abuse, psychosocial and exposure to radiation, and had no specific history of drug allergies and drug use. Contrast-enhanced CT and MRI scans (Figure 1 and Figure 2) revealed a solid mass in close proximity to the liver within the gallbladder exhibiting uneven density and linear separation. Local stenosis affecting the extrahepatic duct along with intrahepatic duct dilatation was also noted. There were no apparent intrahepatic or lymphatic metastases detected. Preoperative laboratory examination indicated mild anemia, AFP levels of 560.60 IU/mL, negative hepatitis B surface antigen results, and negative hepatitis C virus serum tests.
Laparotomy confirmed invasion of tumor cells into the gallbladder chorion and bile ducts in the hepatic hilar region while intraoperative frozen pathological examination showed positive lymph node tumors without explicit intrahepatic metastases present. The patient underwent cholecystectomy, partial liver excision, lymph node dissection at porta hepatis followed by bile duct reconstruction surgery. Post-excision dissection of the tumor displayed excessive bleeding and necrosis. The tumor exhibited fish meat-like appearance with an indistinct boundary between the gallbladder and liver. After 13 days of postoperative treatment, the patient was successfully discharged. Finally, the patient was determined to have gallbladder cancer stage IIIB (according to the UICC/AJCCTNM staging system, 8th edition, 2017).
Histopathological examination of the surgically resected tissue revealed gallbladder hepatoid adenocarcinoma with focal neuroendocrine differentiation due to positive Syn. Positive APF and Hep-1 indicates that the tumor differentiated into hepatocytes, and positive expression of multiple groups of cytokeratin such as CK19, CK 20, CK7, and CK8/18 suggests that the tumor originated from bile duct epithelium (Figure 3 and Figure 4). After obtaining the patient’s informed consent, OrigiMed Company (Shanghai, China) used high-throughput sequencing technology to detect hundreds of genes related to the development and treatment of solid tumors in tumor tissue and blood samples, and conducted PD-L1 immunohistochemical testing, providing comprehensive information on tumor gene variants and PD-L1 protein expression. The results showed the rearrangement of BRCA2 gene, deletions in CDKN2A/B genes, and mutations in TP53 gene.
Two months post-surgery, the patient received a combination therapy of Tegafur, Gimeracil, and Oteracil Potassium capsules (TGO, 100 mg/day, d1–d21) along with Olaparib (600 mg/day), while undergoing regular monitoring of AFP levels and enhanced abdominal CT scans every 2–3 months. The patient tolerated the drug very well and there were no serious adverse reactions that required reducing the dose. Subsequently, over the next 11 months of observation, we found a gradual decline in tumor markers was observed (Figure 5), with no evidence of new masses or suspicious lesions on the CT scans. However, in the 12th month, the patient tested positive for COVID-19 and developed pulmonary infection, necessitating discontinuation of anti-tumor treatment. Unfortunately, lung failure led to the demise of the patient in the 13th month. All events that occur is marked in the axis in Figure 6.
Discussion
Reports of primary hepatocellular adenocarcinoma (HAC) of the gallbladder are exceedingly rare, and there is no consensus on the optimal treatment approach following diagnosis [6]. The prognosis for HAC is extremely poor, with an average survival time reported in the literature of 6.3 (1–20) months [7]. Three cases documented in the literature received postoperative pharmacotherapy, resulting in postoperative survival times of five months (PTX), five months (Gemcitabine and 5-FU), and nine months (Lenvatinib), respectively [4],[7],[8]. Traditional postoperative chemotherapy for gallbladder cancer primarily involves a combination of gemcitabine and platinum drugs; however, it has proven ineffective against HAC. Lenvatinib, as a first-line targeted therapy for liver tumors, exhibits significant inhibitory effects on primary liver cancer; nevertheless, its application in HAC treatment remains controversial [9]. We conducted genetic sequencing analysis on the patient’s tumor tissue and devised a personalized drug treatment plan based on specific genetic variations.
The results demonstrated the presence of BRCA1/2 rearrangement (Level 1A) in the patient, strongly indicating potential efficacy of PARP inhibitors. No gene variations associated with Lenvatinib targets were observed, suggesting ineffectiveness of Lenvatinib for this patient. Furthermore, the patient exhibited CDKN2A/B gene deletions and TP53 gene mutations; however, the evidence level was low (Level 2B). Based on DPYD gene testing, fluoropyrimidine treatment was predicted to offer significant benefits while combining them with platinum drugs would elevate the risk of adverse effects. Therefore, we implemented a treatment regimen comprising Tegafur, Gimeracil, and Oteracil Potassium capsules combined with Olaparib. Following 10 months of drug therapy, progressive reduction in AFP levels and absence of new tumors were observed, confirming treatment effectiveness.
BRCA2 primarily participates in the process of homologous recombination of double-stranded DNA and maintenance of genomic stability. Research has demonstrated that the inactivation of the BRCA2 gene significantly contributes to the development and progression of hepatocellular carcinoma. Individuals with mutations in the BRCA2 allele are at a higher risk for developing malignant liver tumors [10]. The aforementioned statement may serve as the foundation for the occurrence of the patient’s tumor and the efficacy of PARP inhibitor therapy. The TP53 gene is the most commonly mutated tumor suppressor gene in human malignant tumors. Somatic mutations of the TP53 gene have been detected in more than 50% of the tumor genomes, making it the most common mutant gene in tumors and found in a wide range of tumor types. But there are currently no FDA-approved antitumor drugs targeting TP53.
Research on rare malignant tumors such as gallbladder HAC is still in the diagnostic stage, and the efficacy of various treatment methods remains unsatisfactory. The personalized treatment plan we developed for this patient yielded highly favorable outcomes, providing us with invaluable experience in managing this disease entity. However, this case report still has limitations, and we still need larger samples to evaluate the efficacy of genetic testing in the treatment of gallbladder HAC and find clearer potential therapeutic targets. In the future, it is anticipated that there will be an increase in confirmed cases of HAC, and integrating tumor gene sequencing technology offers a more tailored approach for treatment and prognosis assessment of HAC.
In summary, we present a rare case of gallbladder HAC and have devised an effective personalized treatment plan utilizing gene sequencing technology. However, achieving optimal therapeutic strategies for HAC necessitates further accumulation of cases and comprehensive collection of tumor genomic information.
Conclusion
Surgery combined with gene sequencing technology is feasible for treating primary gallbladder HAC, providing new insights for the comprehensive treatment of rare tumors.
REFERENCES
1.
Su JS, Chen YT, Wang RC, Wu CY, Lee SW, Lee TY. Clinicopathological characteristics in the differential diagnosis of hepatoid adenocarcinoma: A literature review. World J Gastroenterol 2013;19(3):321–7. [CrossRef]
[Pubmed]

2.
Ushiku T, Shinozaki-Ushiku A, Maeda D, Morita S, Fukayama M. Distinct expression pattern of claudin-6, a primitive phenotypic tight junction molecule, in germ cell tumours and visceral carcinomas. Histopathology 2012;61(6):1043–56. [CrossRef]
[Pubmed]

3.
Cardinale V, Wang Y, Carpino G, et al. The biliary tree—A reservoir of multipotent stem cells. Nat Rev Gastroenterol Hepatol 2012;9(4):231–40. [CrossRef]
[Pubmed]

4.
Karayiannakis AJ, Kakolyris S, Giatromanolaki A, et al. Hepatoid adenocarcinoma of the gallbladder: Case report and literature review. J Gastrointest Cancer 2012;43 Suppl 1:S139–44. [CrossRef]
[Pubmed]

5.
Li Z, Jiang Q, Chen X, Xiao Y, Xiao J. Primary hepatoid adenocarcinoma of gallbladder with MB21D2/GALNT12/ARID2 mutations: A case report. Front Endocrinol (Lausanne) 2022;12:791153. [CrossRef]
[Pubmed]

6.
Devi NRS, Sathyalakshmi R, Devi J, Lilly SM. Hepatoid adenocarcinoma of the gall bladder—A rare variant. J Clin Diagn Res 2015;9(8):ED09–10. [CrossRef]
[Pubmed]

7.
Iwasaki S, Hidaka H, Uojima H, et al. A case of successful treatment with lenvatinib in primary hepatoid adenocarcinoma of the gallbladder that was difficult to distinguish from hepatocellular carcinoma. Clin J Gastroenterol 2022;15(6):1108–14. [CrossRef]
[Pubmed]

8.
Lee JH, Lee KG, Paik SS, Park HK, Lee KS. Hepatoid adenocarcinoma of the gallbladder with production of alpha-fetoprotein. J Korean Surg Soc 2011;80(6):440–4. [CrossRef]
[Pubmed]

9.
Gordan JD, Kennedy EB, Abou-Alfa GK, et al. Systemic therapy for advanced hepatocellular carcinoma: ASCO guideline update. J Clin Oncol 2024;42(15):1830–50. [CrossRef]
[Pubmed]

10.
Katagiri T, Nakamura Y, Miki Y. Mutations in the BRCA2 gene in hepatocellular carcinomas. Cancer Res 1996;56(20):4575–7.
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Miao Tang - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Zeqiang Hu - Acquisition of data, Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Hejun Gao - Acquisition of data, Analysis of data, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was obtained from the patient for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2025 Miao Tang et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.