|
Case Report
First report of robotic-assisted partial gastrectomy of a large lymphatic-venous malformation in a teenager: A case report
1 MD, Pediatric Surgery Research Fellow, Department of Surgery, Loma Linda University Health, Loma Linda, CA, USA
2 MD, Assistant Professor, Division of Pediatric Surgery, Department of Surgery, University of Florida, Gainesville, FL, USA
3 MD, Pediatric Surgery Research Associate, Department of Surgery, Loma Linda University Health, Loma Linda, CA, USA
4 MD, PhD, Associate Professor, Division of Pediatric Surgery, Department of Surgery, Loma Linda University Health, Loma Linda, CA, USA
5 MD, Adjunct Clinical Assistant Professor, Department of Surgery, Indiana University, Indianapolis, IN, USA
6 MD, MS, MBA, Professor, Division of Pediatric Surgery, Department of Surgery, Loma Linda University Health, Loma Linda, CA, USA
Address correspondence to:
Joyce McRae
MD, 11175 Campus Street, CP21111, Loma Linda, CA 92350,
USA
Message to Corresponding Author
Article ID: 100166Z12JM2026
Access full text article on other devices

Access PDF of article on other devices

How to cite this article
McRae J, Raymond SL, Minhas A, Radulescu A, Srikureja D, Tagge E. First report of robotic-assisted partial gastrectomy of a large lymphatic-venous malformation in a teenager: A case report. J Case Rep Images Surg 2026;12(1):5–9.ABSTRACT
Introduction: Gastrointestinal vascular anomalies are rare in children. A subtype, lymphatic-venous malformations, is even rarer. These anomalies can result in serious sequelae such as bleeding. Management of these rare pathologies is challenging and may involve endoscopic, angiographic, or surgical management.
Case Report: We present the case of a 14-year-old female who presented to the hospital after hematemesis and was found to have a gastric vascular malformation. Workup included cross-sectional imaging, endoscopy, and angiography. After multidisciplinary discussion, definitive management was achieved with a robotic-assisted partial gastrectomy with longitudinal gastric closure. The surgical pathology was diagnostic of a lymphatic-venous malformation. No complications occurred in the perioperative period through 10 months of follow-up.
Conclusion: This is the first report of a robotic-assisted partial gastrectomy with supplemental use of indocyanine green and endoscopy for gastric lymphatic-venous malformation. Use of the robotic platform for minimally invasive resection is safe and can be considered for management of this rare pathology.
Keywords: Gastric lymphatic-venous malformation, Pediatric indocyanine green, Pediatric robotic-assisted partial gastrectomy, Pediatric vascular malformation
Introduction
Gastrointestinal (GI) vascular anomalies are exceedingly rare in children, accounting for a small fraction of GI bleeding etiologies [1]. An accurate prevalence is difficult to determine in children as they are often discovered only when a patient is being evaluated for a gastrointestinal bleed [2]. Pediatric gastric vascular anomalies, in particular, pose significant therapeutic challenges, especially if the malformation involves adjacent structures such as the esophagus or spleen [3]. We present a case of a pediatric gastric lymphatic-venous malformation (LVM) not amenable to endoscopic or other noninvasive angio-ablation methodologies. We describe the first pediatric case of a large gastric LVM managed successfully with robotic-assisted resection.
Case Report
A previously healthy 14-year-old female presented to the hospital after a syncopal event associated with generalized weakness, vague abdominal discomfort, nausea, and bloody emesis. Prior to presentation, the patient had a normal appetite and denied any abnormal stools. Initial vital signs included tachycardia without hypotension. Physical exam was notable for pallor but no abnormal abdominal findings.
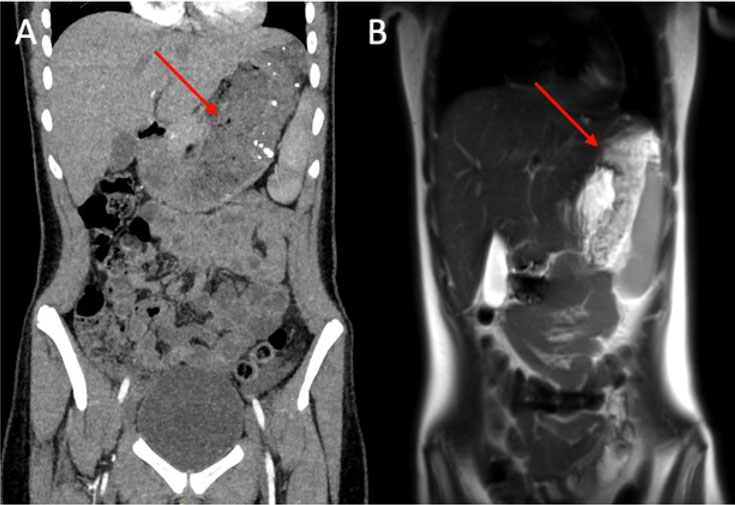
A complete blood count (CBC) demonstrated a hemoglobin of 3.5 g/dL, hematocrit of 14%, white blood cell (WBC) count of 16.9 bil/L and normal platelet count. Due to her initial critically ill presentation, she was appropriately resuscitated with blood products, and after stabilization, underwent additional evaluation to identify the source of bleeding. Contrast-enhanced computed tomography abdomen/pelvis (CTAP) demonstrated an irregular thickening of the stomach measuring up to 1.9 cm with enhancement of the lateral-posterior gastric wall and a subtle focus of venous and arterial enhancement. A magnetic resonance imaging (MRI) of the abdomen with contrast confirmed lateral-posterior wall thickening of the stomach, measuring up to 2.4 cm in thickness, with extension into the adjacent omentum and spleen. The imaging differential diagnoses included malignancy versus vascular anomaly (Figure 1).
Esophagogastroduodenoscopy (EGD) demonstrated a large 13 cm × 6 cm, multilobular vascular lesion in the proximal body of the stomach, greater curvature, and fundus. The duodenum was normal. Biopsies were non-diagnostic demonstrating normal duodenal mucosa with reactive gastric and esophageal mucosa. Endoscopic ultrasound (EUS) revealed internal flow seen on Doppler. Upon completion of the endoscopy active bleeding was noted, consistent with the friable nature of the lesion. Due to hypotension, the patient was taken emergently to Interventional Radiology (IR) for possible embolization; however, celiac angiography showed no active extravasation, no intervention was performed and the patient was stabilized with medication administration.
Subsequently, her case was presented at the Vascular Anomalies Center multidisciplinary conference. Given the lesion’s size, vascularity and proximity to the spleen, they recommended open surgical management over endoscopic or IR interventions. A local multidisciplinary discussion led by Pediatric Surgery with input from IR and Minimally Invasive Surgery (MIS) resulted in the decision to proceed with surgical intervention.
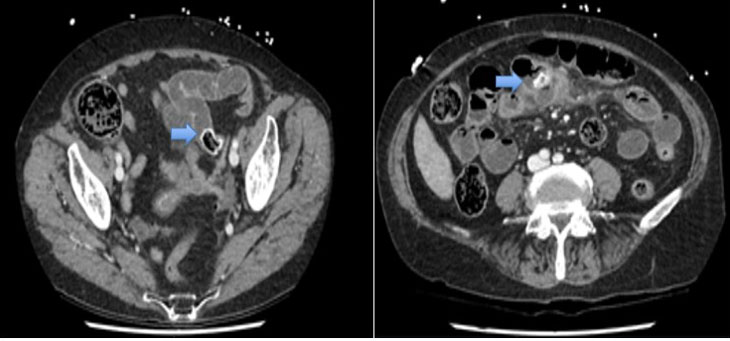
A robotic-assisted partial gastrectomy, omentectomy and EGD during the index admission was planned. After insufflation and standard robotic access, an EGD confirmed malformed gastric mucosa (Figure 2A). The endoscope was left in place to assist with the dissection. Indocyanine green (ICG) angiography showed no arterial-phase fluorescence within the vascular anomaly but highlighted surrounding tissues, delineating its borders (Figure 2B). Our ICG protocol included 5 mg ICG injection followed immediately by 10 cc saline fast flush, with neutral field visualization and visualization of ingress.
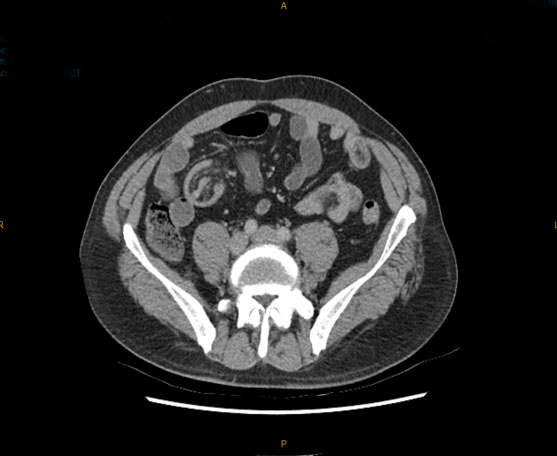
The mass extended into the omentum and gastrosplenic fat, involving the short gastric arteries (Figure 3A). This necessitated a wider dissection, achieved with suture ligation, clips and a vessel sealer to ensure hemostasis. Robotic dissection continued to the left crus, providing excellent exposure of the vascular anomaly involving the greater curve. Posterior attachments to the pancreas were divided, and the vascular anomaly was isolated. We elected to devascularize the lesion by dividing the stomach around it to minimize bleeding. Using a vessel sealer, the anterior portion of the vascular anomaly was excised (Figure 3B). This facilitated circumferential excision of the lesion. Reconstruction was performed with the endoscope as a bougie along the lesser curve, guiding linear closure (Figure 3C). Longitudinal gastric closure was completed with a running full thickness layer of 2-0 Vicryl followed by a seromuscular running layer of 2-0 Stratafix from cardia to angularis with no noted narrowing at the angularis or antrum on completion (Figure 3D). An air insufflation leak test was negative, and a drain was placed in the lesser sac. Total console time was 199 minutes.
The patient required no intraoperative or postoperative transfusions. On postoperative day 3, a fluoroscopic upper gastrointestinal (UGI) study showed no leak. A small-volume, sugar-free, non-carbonated clear liquid diet was initiated and advanced to full liquids within two days. She was discharged home on postoperative day 6 on a bariatric full liquid diet, with the drain removed.
Pathology confirmed a large vascular malformation involving the mucosa, submucosa, muscularis, and peri-gastric soft tissue and omentum. It was composed predominantly of venous and lymphatic channels with a minor arterial component, diagnostic of LVM.
She advanced to a regular diet at six weeks. She remained asymptomatic during 10 months in the outpatient setting but was lost to long-term follow-up.
Discussion
Vascular malformations are rare pathologies with limited understanding of their pathogenesis [4]. They are often seen in the head and neck and are associated with genetic conditions such as hereditary hemorrhagic telangiectasia and capillary malformation-arteriovenous malformation [4]. The predominant hypothesis of origin is that vascular malformations arise from errors in vascular morphogenesis, resulting in abnormal connections between arteries and veins that bypass the capillary bed [5]. The International Society for the Study of Vascular Anomalies (ISSVA) Classification for Vascular Anomalies organizes vascular anomalies into vascular tumors, vascular malformations or potentially unique vascular anomalies [6]. The ISSVA further delineates malformations by flow state and major component [6].
Management of symptomatic pediatric gastrointestinal vascular anomalies can be challenging. Endoscopic therapies (argon plasm coagulation, laser ablation, or mechanical resection), and pharmacologic approaches (e.g., angiogenesis inhibitors) have been described, but definitive treatment often requires surgical resection [1],[7],[8]. Surgical resection of vascular malformations focuses on resection of all grossly abnormal tissue, unless the lesion is large and in a sensitive location benefitting from attempts at a stepwise size reduction with preoperative embolization and sclerotherapy [9]. Decreasing the size of the lesion if feasible preoperatively should decrease the risk for major blood loss [9]. The robotic platform has been previously safely utilized for the management of adult vascular anomalies in the lung, mediastinum and pelvis [10],[11],[12]. To date, robotic partial gastrectomy has not previously been reported as a management strategy for this condition in children or adults.
Robotic surgical technology has evolved since its inception in adults in 1997 and its first use in children in 2002 [13],[14]. In comparison to traditional laparoscopic gastrectomy, robotic-assisted gastrectomy in adults has recently been found to have fewer postoperative complications, less estimated blood loss, faster postoperative recovery, and lower conversion rate [15]. Its advantages—three-dimensional field of view, magnification, improved dexterity, tremor filtration, and consistent retraction—are particularly valuable in complex gastric resections [16]. Lesions of the gastric fundus and cardia are technically demanding to manage laparoscopically due to limited access, angulation, and suturing constraints. The robotic platform mitigated these challenges in our patient, enabling safe dissection and layered closure, and facilitated use of ICG to delineate the lesion’s margins. With the integration of artificial intelligence into medical care, the future of managing this condition robotically may integrate imaging overlay for additional surgical decision-making support.
Conclusion
This case highlights a rare large pediatric gastric LVM involving the overlying omentum. The robotic platform and the use of ICG facilitated the identification of the borders of the mass and provided technical advantages that enabled safe resection and reconstruction in a challenging region. To our knowledge, this represents the first documented pediatric case of robotic-assisted partial gastrectomy for a giant gastric LVM, demonstrating its feasibility and utility.
REFERENCES
1.
Würfel C, Brückner S, Aust DE, Straub S, Hauck F, Laass MW. Intestinal microvascular malformations and congenital asplenia in an adolescent possibly expanding the phenotype of Ivemark syndrome. Eur J Gastroenterol Hepatol 2011;23(12):1258–61. [CrossRef]
[Pubmed]

2.
Irish MS, Caty MG, Azizkhan RG. Bleeding in children caused by gastrointestinal vascular lesions. Semin Pediatr Surg 1999;8(4):210–3. [CrossRef]
[Pubmed]

3.
Schettini ST, Ribeiro RC, Brito PL, Abib Sde CV, Onofre LS, Kawasaki LH, et al. Gastric hemangioma in a 5-year-old boy. J Pediatr Surg 2007;42(4):717–8. [CrossRef]
[Pubmed]

4.
Schimmel K, Ali MK, Tan SY, Teng J, Do HM, Steinberg GK, et al. Arteriovenous malformations-current understanding of the pathogenesis with implications for treatment. Int J Mol Sci 2021;22(16):9037. [CrossRef]
[Pubmed]

5.
Fishman SJ, Burrows PE, Leichtner AM, Mulliken JB. Gastrointestinal manifestations of vascular anomalies in childhood: Varied etiologies require multiple therapeutic modalities. J Pediatr Surg 1998;33(7):1163–7. [CrossRef]
[Pubmed]

6.
International Society for the Study of Vascular Anomalies (ISSVA), editor. ISSVA Classification & Glossary for Vascular Anomalies by International Society for the Study of Vascular Anomalies (2025). ISSVA World Congress; 2025; Madrid, Spain. [Available at: https://www.issva.org/classification].

7.
López-Gutiérrez JC. Hemangiomas and vascular malformations of the stomach. J Pediatr Surg 2007;42(9):1634–5; author reply 1635–6. [CrossRef]
[Pubmed]

8.
Pedersen FM, Vilmann P, Bytzer P. Gastric arteriovenous malformation: Doppler EUS-guided diagnosis and therapy. Gastrointest Endosc 2002;55(4):597–9. [CrossRef]
[Pubmed]

9.
Johnson AB, Richter GT. Surgical considerations in vascular malformations. Tech Vasc Interv Radiol 2019;22(4):100635. [CrossRef]
[Pubmed]

10.
Lengyel BC, Watson JB, Kim MP, Lumsden AB. Robotic right lower lobectomy for a persistent large pulmonary arteriovenous malformation following repeated coil embolization. J Vasc Surg Cases Innov Tech 2024;10(6):101605. [CrossRef]
[Pubmed]

11.
Sasankan P, Geraci TC, Narula N, Cerfolio R. Robotic resection of a combined capillary and arteriovenous malformation in the mediastinum. Ann Thorac Surg 2021;111(3):e189–91. [CrossRef]
[Pubmed]

12.
Johansson C, Chan F. Robotic-assisted resection of pelvic arteriovenous malformation utilising intraoperative video angiography with indocyanine green. Journal of Minimally Invasive Gynecology 2022;29(11 Suppl):S37. [CrossRef]

13.
Himpens J, Leman G, Cadiere GB. Telesurgical laparoscopic cholecystectomy. Surg Endosc 1998;12(8):1091. [CrossRef]
[Pubmed]

14.
Gutt CN, Markus B, Kim ZG, Meininger D, Brinkmann L, Heller K. Early experiences of robotic surgery in children. Surg Endosc 2002;16(7):1083–6. [CrossRef]
[Pubmed]

15.
Li Z, Zhou W, Yang W, Miao Y, Zhang Y, Duan L, et al. Efficacy and safety of robotic vs. laparoscopic gastrectomy for patients with gastric cancer: Systematic review and meta-analysis. Int J Surg 2024;110(12):8045–56. [CrossRef]
[Pubmed]

16.
Jacobson JC, Pandya SR. Pediatric robotic surgery: An overview. Semin Pediatr Surg 2023;32(1):151255. [CrossRef]
[Pubmed]

SUPPORTING INFORMATION
Author Contributions
Joyce McRae - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Steven L Raymond - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Amna Minhas - Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Andrei Radulescu - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Daniel Srikureja - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Edward Tagge - Conception of the work, Design of the work, Acquisition of data, Analysis of data, Drafting the work, Revising the work critically for important intellectual content, Final approval of the version to be published, Agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
AcknowledgmentsArtificial Intelligence was not utilized in the creation or development of this work.
Guarantor of SubmissionThe corresponding author is the guarantor of submission.
Source of SupportNone
Consent StatementWritten informed consent was waived by Institutional Review Board for publication of this article.
Data AvailabilityAll relevant data are within the paper and its Supporting Information files.
Conflict of InterestAuthors declare no conflict of interest.
Copyright© 2026 Joyce McRae et al. This article is distributed under the terms of Creative Commons Attribution License which permits unrestricted use, distribution and reproduction in any medium provided the original author(s) and original publisher are properly credited. Please see the copyright policy on the journal website for more information.